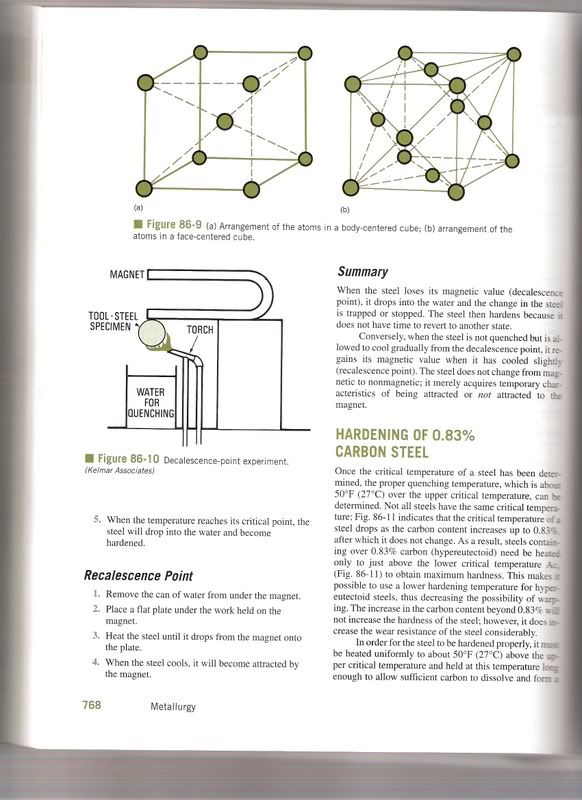
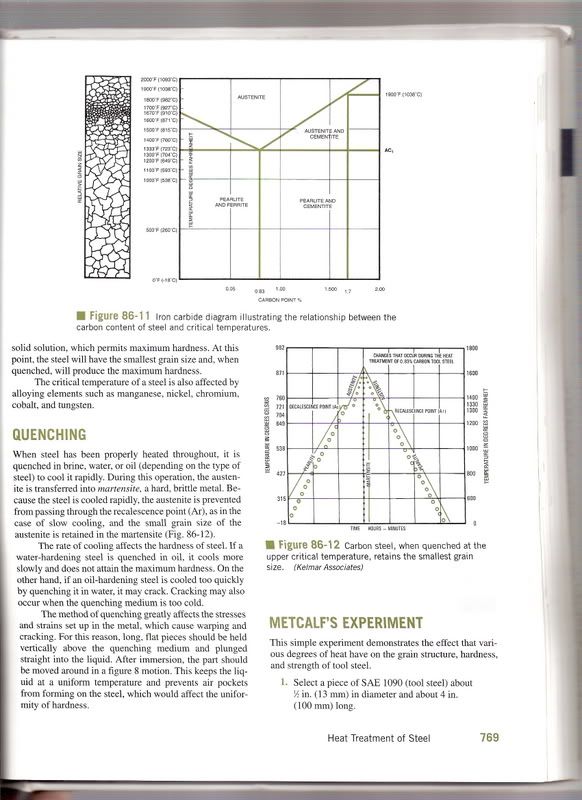
This is absolute BS. Cementite (or Iron Carbide Fe3C) forms in steel and iron as it is cooled below its lower critical temperature, which is around 725 Celsius. Above this temperature, carbon is actually solulable in iron, but when it cools, it is "kicked out" of solution with the iron, and coalesces into pockets of cementite and almost pure iron, called ferrite. Together these patches of ferrite and cementite are termed "Pearlite". These microstructures are here to stay, and in any particular proportion that they were originally present in. They can be modified by cold working, forging, or heat treating, but not changed in quantity. One cannot form more cementite in a piece of iron, no matter how hard you brake.
I'd love to scan my notes and textbooks on the subject, if anyone disagrees.
I studied metallurgy in school, being, well, a machinist, so if you want to argue about metal, you've come to the wrong place.
Oh, and who the hell is Carroll Smith?
Finally, who honestly believes that a brake rotor that is heated unevenly and cooled even more unevenly is not going to experience internal stresses leading to warping and cracking? Think about it. The outer disc of the rotor is heated rapidly, as well as subjected to a lot of force, while the center of the rotor sits on a nice cold hub. Furthermore, that smoking hot rotor starts to cool as soon as you do stop, well, except for the 1/3 of its face which is still covered by and in contact with smoking hot brake pads. Now it is cooling unevenly across its face. Tell me that won't warp a rotor.
Cementite indeed!








 Rotors on those cars unfortunately cost upwards of $300 a set.
Rotors on those cars unfortunately cost upwards of $300 a set.






Bookmarks